Heparin
Purpose
We offer method development, method validation and structural characterization study of many different sulfated products including, heparin, low-molecular-weight heparins (LMWHs), synthetic heparins analogues to evaluate active ingredient sameness of the generic and innovator products.
Services Offered
Equivalence of physicochemical properties
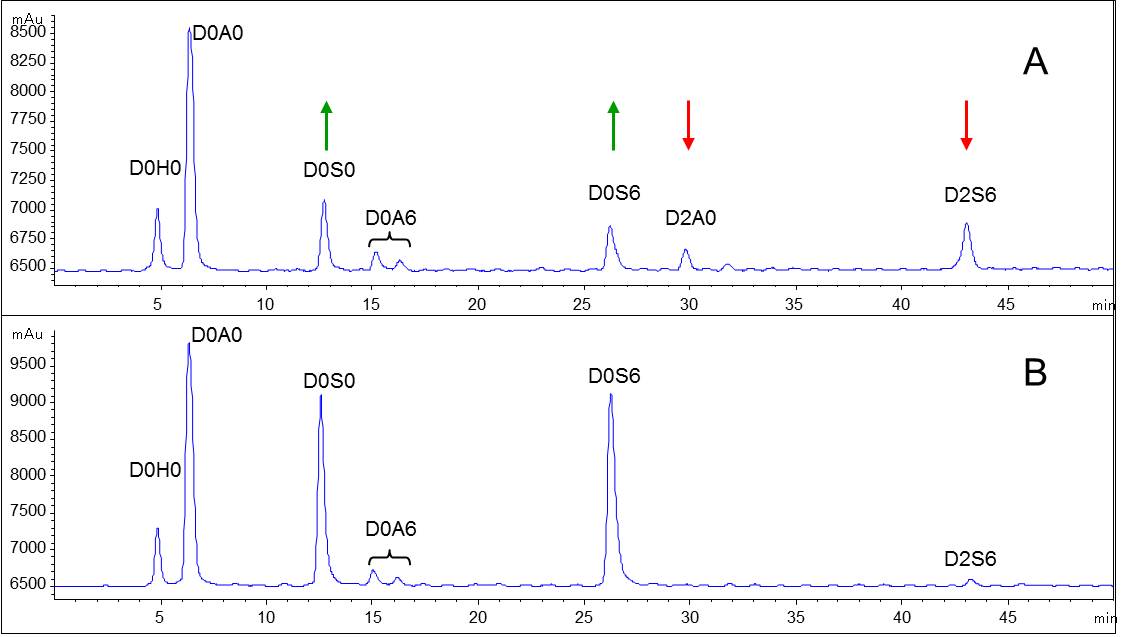
We apply size exclusion chromatography (SEC), liquid chromatography-mass spectrometry (LC-MS), NMR, as other techniques to obtain molecular weight parameters and characteristic fingerprints for comparison of overall composition of biosimilars.
Equivalence of source material and mode of depolymerization
Methods for source material characterization include strong anion exchange chromatography (SAX-HPLC), high pH anion exchange chromatography (HPAEC), and NMR.
Equivalence of building blocks, fragment mapping, sequence and linkage of oligosaccharide species
We perform detailed, bottom-up analysis of complex mixtures by NMR, SAX-HPLC, and LC-MS.