Routine Services
Glycan Profiling of N and O-Linked Oligosaccharide Profiling from Animal, Plant and Bacteria
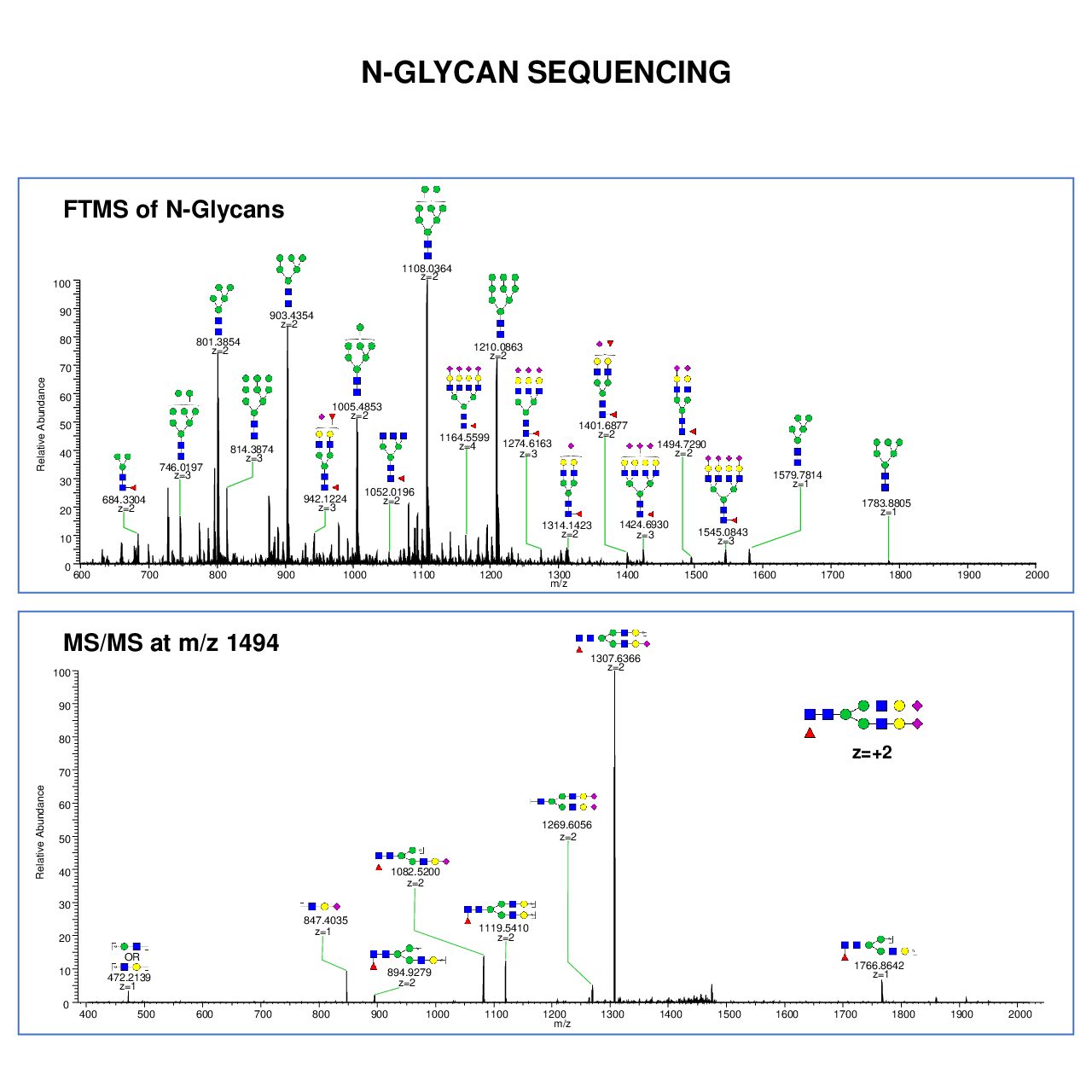
In glycan profiling we can release, purify and derivate of N and O-linked -glycans by enzymatic or chemical methods such as Beta-elimination and hydrazinolysis prior to MS Profiling. We can start from as small as 50μg of starting protein. The glycans can be prepared from a single purified glycoprotein or mixed protein and also from cells, tissues, culture supernatants, and gel slices.
We perform compositional analysis and sequencing of N- and O-linked glycans after release and permethylation, as well as glycan profiling of N and O-linked oligosaccharide profiling from animal, plant and bacteria. We can release, purify and derivate of N and O-linked glycans by enzymatic (PNGase F or A) or chemical methods such as Beta-elimination and hydrazinolysis prior to MS profiling.
- Full characterization of a target protein’s glycosylation includes N– and O-glycoform structures, mapping of glycosylation sites and extent of glycosylation at each site.
- Analysis of peptides and glycopeptides produced following protease digestion is readily achieved by LC-ESI-MS/MS or nano-LC-NSI-MS/MS with the CCRC Analytical Service’s Thermo LTQ- Fusion Tribid and LTQ-Velos instrumentation.
- Electron transfer dissociation (ETD) and high collision dissociation (HCD) fragmentation capabilities enhance glycopeptide analysis and site of glycosylation studies.
- While most glycopeptide analyses are completed by manual data interpretation, numerous software such as Thermo Proteome Discoverer™ and Protein Metric’s Byonic ™ are used supplementary for our data analysis.
For more in depth analysis please go to https://ast.uga.edu/glycomicsglycoproteomics/