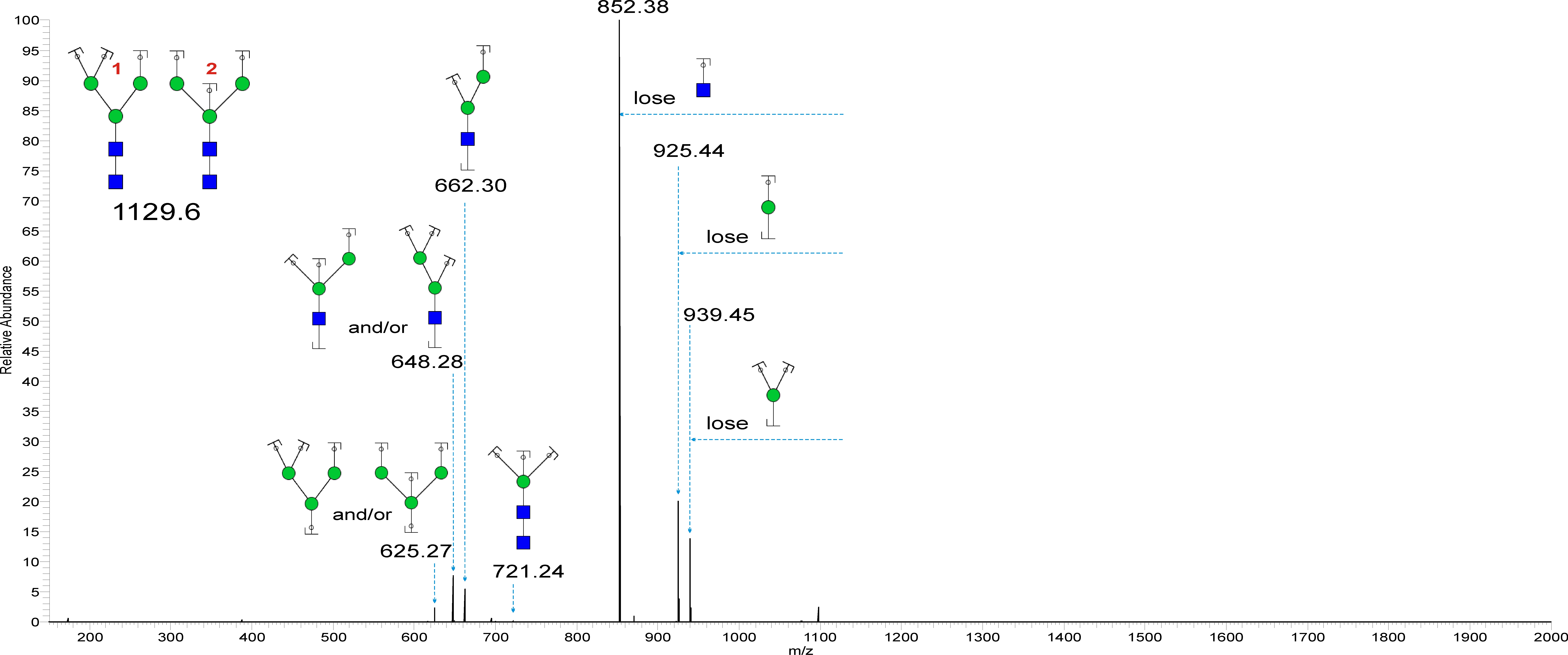
Glycoprotein Structure Analysis
We compare antibody and glycoprotein innovator drug products (reference listed drugs) with generic active pharmaceutical ingredients (API).
Services Offered
Glycosyl quantitative composition
The products are analyzed by glycomics and glycoproteomics approaches, including N- and O-linked glycan profiling, glycopeptide analysis, glycosylation site mapping, and oligosaccharide heterogeneity (glycoforms) as well as identification of isomers (isobaric structures).
Glycan linkage analysis
Linkage analysis of polysaccharides and oligosaccharides is performed by partially methylated alditol acetate (PMAA) method to determine the linkage of monosaccharides and branching points.
Glycopeptide analysis
- Peptide mapping using enzyme digestion and mass spectrometry (MALDI-MS, LC-MS, and LC-NSI -MS/MS) including N– and C– terminal sequences.
- Disulphide bridge mapping and chemical modifications by MS.
- Identification of Post Translational Modifications (PTMs) e.g. Deamidation, Acetylation, Phosphorylation, Sulfation.
- 1D and 2 D NMR for 3D Structure.
- Identification and quantitation of Fatty acids